Learn about the CHOICES and ReCode trials
Have a listen to Prof Kors van der Ent, the project’s principal investigator, and other members of the research team in our new video. For the latest updates, make sure to read our newsletters!
What is HIT-CF Europe?
HIT-CF Europe is a research project which aims to provide better treatment and better lives for people with cystic fibrosis (CF) and rare mutations. To achieve this, drug candidates of several companies will first be tested in the laboratory on patient-derived mini-intestines (organoids). Secondly, based on the reaction in the organoids, a smaller group of patients will be assigned to studies (clinical trials) with one of the drug candidates.
All participating centres are part of the ECFS-CTN.
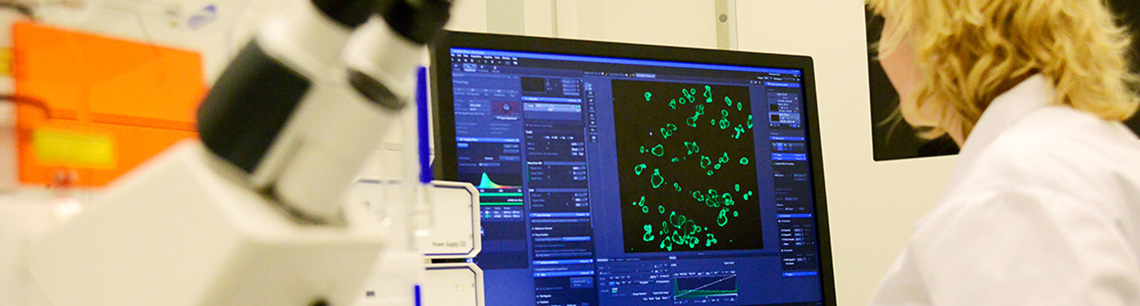
What is the use of organoids?
Organoids are cell cultures that grow in a culture dish, and look similar to the organ from which they are derived. Intestinal organoids can therefore also be called mini-intestines. To make intestinal organoids for the HIT-CF Europe project, rectal tissue samples (biopsies) will be obtained. This procedure is not painful and will take 5-10 minutes. Because organoids are made from stem cells, they contain the same mutations as the person from whom the biopsies are derived. The drug candidates target the basic defect of CF, and the organoids will be used to test on which mutations the drugs have a positive effect.
-
1
We take some tissue from the intestine
-
2
This tissue is cultured in the lab
-
3
The result is an organoid
-
4
On this organoid we test medicines
-
5
And we bring the correct medicines to the patients
Pushing forward
HIT-CF makes sure people with rare mutations are not left behind in the development of new treatments. This video explains how we are pushing forward.
Towards personalized medicine
New mutation class-specific drugs are currently only being clinically tested in patients with well-described, very common mutations. As a result, market authorization and reimbursement of these drugs is only granted in these specific subsets of patients. Nevertheless, other patients with less common mutations might also benefit from them. The ultimate goal of HIT-CF Europe is to develop a path for access to therapies for individual patients or patient groups who show positive response to the therapy in an organoid test and pave the way for organoid-based personalized medicine.
Share this page:














